Key Features
SmartTilt Technology
SmartTilt ensures that the Brilliance Pro works effectively at any position and along with other pieces of neonatal intensive care equipment. This technology provides light of uniform intensity across the treatment area.
Energy Efficient
Consumes less than half the power consumed by CFLs.
Minimal Maintenance
LEDs last 60 times longer compared with CFLs. Hospitals can enjoy considerable savings by avoiding costly bulb replacements.
Quality
Designed to withstand severe power fluctuations without any change in performance.
Therapy Timer
Treatment times are easily tracked and readings are displayed on an LCD screen.
Effective Surface Area
Maximum possible body surface area for effective phototherapy as per the recommended standards of American Academy of Pediatrics.
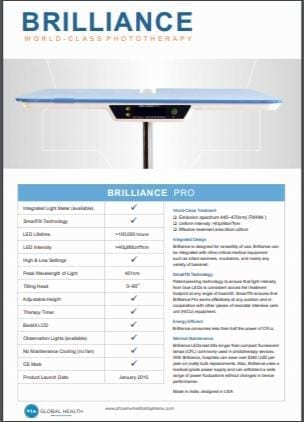
Specifications
- LED Lifetime: >100,000 hours
- LED Intensity: >40µW/cm²/nm
- Peak Wavelength of Light: 451nm
- Electrical Supply (Voltage, Frequency): 100 – 240 VAC – 50/60Hz (medical grade)
- Maximum Power Consumption: 42W
- Illumination Source: Blue LEDs and white observation lights
- Peak Spectral Irradiance: >40µW/cm²/nm at recommended treatment distance measured with Ocean Optics Spectrophotometer
- Emission Spectrum: 445-470 nm (bounds of FWHM range)
- Effective Surface Area: 50 x 25 cm (at recommended treatment distance)
- Irradiance Ratio: >0.4 (minimum/maximum irradiance)
- Time Totalizer: Machine run time totalizer plus therapy time totalizer
- LCD: Backlit
Range
- Tilting Head: 0-90 degrees
- Height Adjustment Range: 118-165 cm (distance between bottom surface and ground)
Dimensions and Weight
- Product Dimensions: 123 x 69 x 64 cm
- Light Unit Dimensions: 55 x 30 x 5 cm
- Total Unit Mass: 12kg
- Footprint of Stand: 69 x 64 x 10 cm
- Height of Base: <10 cm
- Castors: 2 in, lockable
Use
- Recommended Treatment Distance: 45 cm
Standard Accessories
Not applicable
Optional Accessories & Add-ons
Not applicable
Product Documents, Manuals & Brochures
Data Sheet
| Ghana, India, Kenya, Nigeria, Tanzania |
Regulatory Certificates

ISO 13485 Certificate

CE Certificate
Videos
Not available at this time
Frequently Asked Questions
Not available at this time
Studies & Trials
Not available at this time
Package Contents
- 1x Brilliance Pro Double Surface Phototherapy Unit: including both top and bottom phototherapy lights and built-in infant bed
Warranty
This warranty covers repair/replacement of any defective hardware items, contingent to these defects having arisen only out of defective material or workmanship. If brought to the notice of Phoenix Medical Systems (P) Ltd in writing on or before the date of expiry of the warranty mentioned on the warranty card. The warranty is applicable for a period of 12 months from the date of commissioning or 13 months from the date of dispatch from Phoenix Medical Systems (P) Ltd, whichever is earlier. The parts or products not manufactured by Phoenix Medical Systems (P) Ltd but supplied as a part of an equipment shall be covered by this warranty.
The warranty does not cover:
Misuse, negligence, accidents and improper handling. Improper installation or maintenance practice or any alteration carried out on the equipment. Wear and tear occurring due to normal usage.
Testimonials
Not available at this time